Carbon Dioxide State At Room Temperature

An aqueous solution turns litmus from blue to pink.
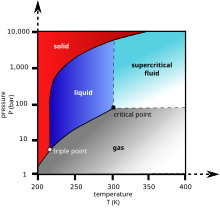
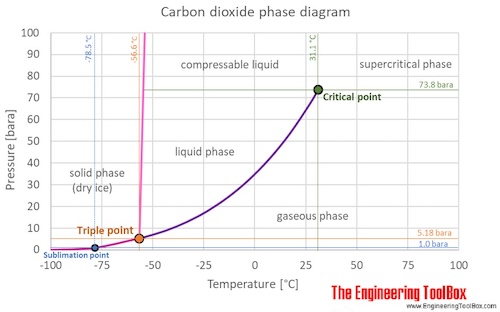
Carbon dioxide state at room temperature. Dry ice is the solid form of carbon dioxide. Structure and properties. When the carbon dioxide concentration goes down temperature goes down. It can only exist at a pressure above 5 1 atm 5 2 bar.
Liquid carbon dioxide is the liquid state of carbon dioxide which cannot occur under atmospheric pressure. To convert heat values to joules per mole values multiply by 44 095 g mol. At 1 atm it is a solid at temperatures below 78 c. Carbon dioxide co2 cid 280 structure chemical names physical and chemical properties classification patents literature biological activities safety.
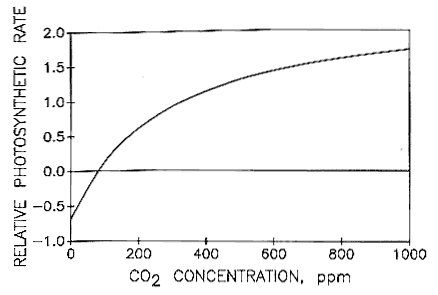
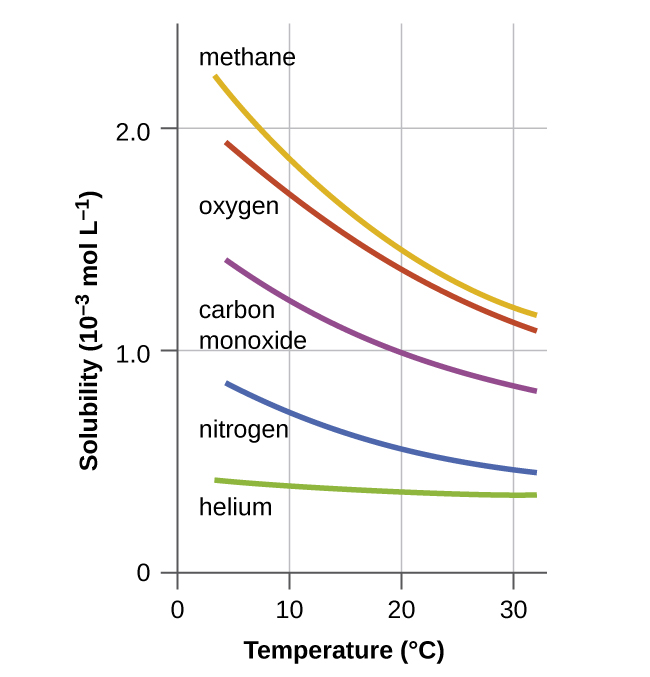
Carbon dioxide at room temperature is a gas. Heat content data heat of vaporization and entropy values are relative to the liquid state at 0 c temperature and 3483 kpa pressure. In its solid state carbon dioxide is commonly called dry ice. A small part of the correspondence is due to the relationship between temperature and the solubility of carbon dioxide in the surface ocean but the majority of the correspondence is consistent with a feedback between carbon dioxide and climate.
Co 2 is an acidic oxide. Carbon dioxide has no liquid state at pressures below 5 1 standard atmospheres 520 kpa. Dry ice will sublime or turn from a solid state into a gas state at room temperature. If you left dry ice.
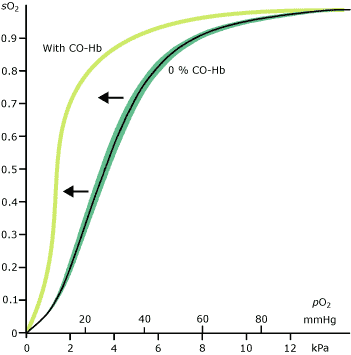
The answer to your question is in the molecular structure of co2 there are 4 electrons in outer orbit of carbon and six in that of oxygen. Co 2 in concentrations of 7 to 10 cause dizziness headache visual and hearing dysfunction and unconsciousness within a few minutes to an hour. Msds for solid carbon dioxide is available from pacific dry ice inc. At 1 atmosphere near mean sea level pressure the gas deposits directly to a solid at temperatures below 78 5 c 109 3 f.
194 7 k and the solid sublimes directly to a gas above 78 5 c.