Carbonated Sparkling Water Solute And Solvent

Seltzer water sparkling water.
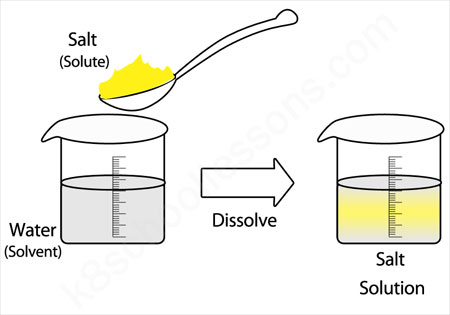
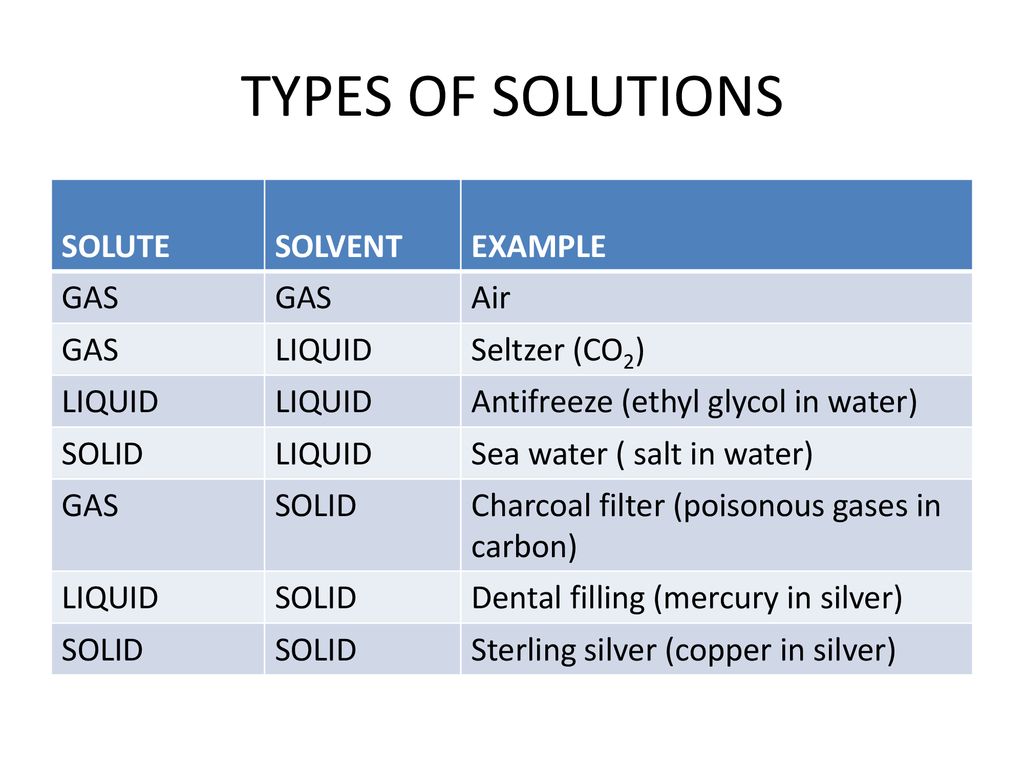
Carbonated sparkling water solute and solvent. Sparkling water is also known as club soda or seltzer water. In this type of solution the solute is a and the solvent is a. Sparkling water is a carbonated water. Carbon dioxide gas was dissolved under pressure before adding to the water.
A mixture in which the particles of solute in a solvent are so large that they settle out unless the mixture is constantly stirred or agitated types of solutions solutions exist in phases of solids liquids and gases depending on the temperature and pressure. The beverage might also. Carbonated water is water that has been infused with carbon dioxide gas under pressure. The best answer i could tell you is that the solute would be the carbonated water and the solvent would be the salt.
Which of the following best explains why covalent solutions have poor electrical conductivity. When you mix soda and salt the solute and the solvent would be a variety of things. Carbon dioxide is the gaseous solute and water is the liquid solvent the carbon dioxide gas ivess the drink its fizz. Filtered sparkling water systems and dispensers found in offices restaurants hotels and resorts typically use this method to ensure the water they are offering to patrons staff and guests is.
Name the solute and solvent in the following. Carbonated beverages are liquid gas solutions. When you mix soda and salt the solute and the solvent would be a variety of things. This produces a bubbly drink that s also known as sparkling water club soda soda water seltzer water.
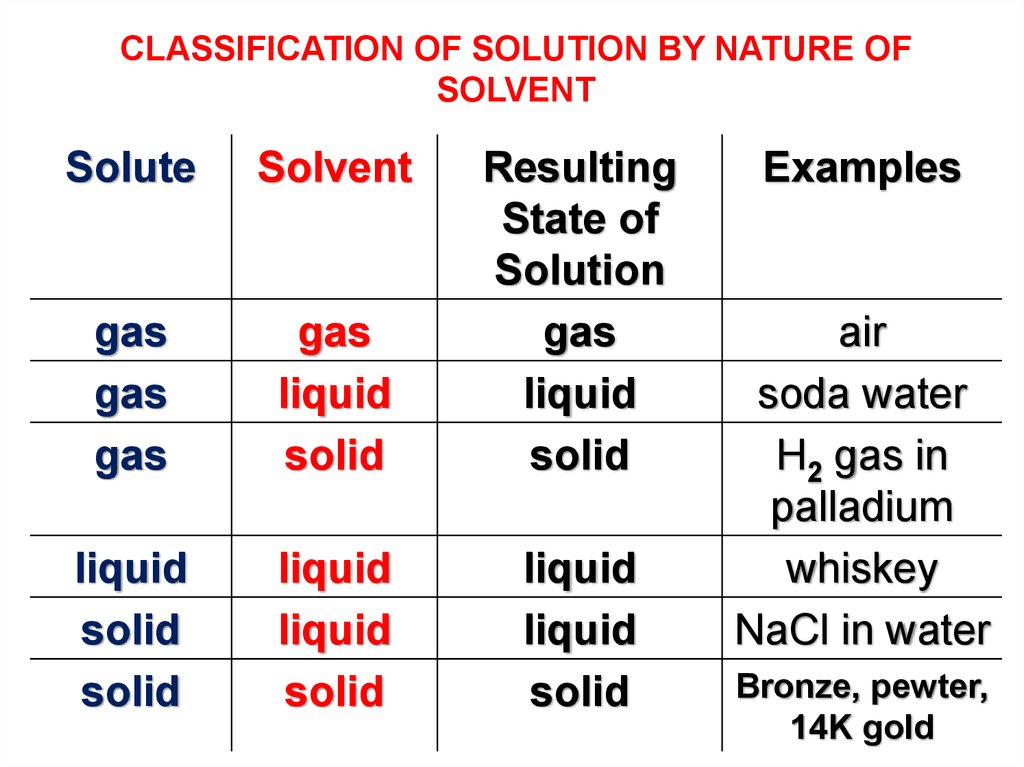
This bubbly water is pure and simple. Carbonated beverage o2 in h2o swimming pool cl2 in h2o air o2 in n2. Attractive forces between solute and solvent must be greater than solute solute attractive forces. The best answer i could tell you is that the solute would be the carbonated water and the solvent would be the salt.